Androgen Receptor Mediated Gene Regulation
About this project
Project information
Project status
In progress
Contact
Research subject
Research environments
Androgens mediate their physiological function through interactions with receptors belonging to the steroid hormone receptor super-family. The receptors activate different genes by binding as homodimers to a variety of different gene response elements. The genes for the receptors and the binding proteins are highly conserved between species and show specific expression patterns during development as well as in different tissues.
We are interested in the conservation of ligand-binding characteristics of androgen receptors from different species and how the difference in amino acid sequence is translated into species-specific responses. Our focus is on the comparison of receptors from mammals, birds and fish. There is a general difference in androgen usage in different species with 5α-dihydrotestosterone being the most potent ligand in mammals while testosterone is the preferred ligand in birds. In many fish species 11keto-testosterone is the most potent androgen. This difference in ligand utilization between these classes of animal suggests that they will show differential responses to man made chemicals with androgenic or anti-androgenic potential.
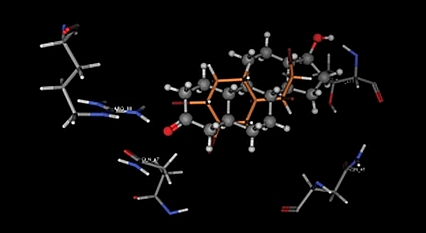
We are using molecular modeling in combination with experimental analysis of receptor activation in order to identify novel androgenic and anti-androgenic compounds. So far we have identified one brominated flame retardant, TBECH (1,2-dibromo-4-(1,2 dibromoethyl)cyclohexane or tetrabromoethylcyclohexane) as a potent androgenic substance. The findings have been reported in the Journal of Medicinal Chemistry and in Environmental Health Perspectives.
Ongoing projects
1. Identification of novel androgenic and anti-androgenic brominated compounds using modeling, receptor activation and gene expression assays. Includes studies on human, chicken and zebrafish androgen receptors.
2. Evolutionary conservation of the three-spined stickleback androgen regulated gene, Spiggin.
3. Identification of gene mutations in androgen receptors leading to altered ligand recognition, with potential importance for cancer development.
4. Optimization of computer modeling. Can we define criteria for ligand-receptor interaction and agonist/antagonist action.
Selected publications
Asnake S, Pradhan A, Banjop-Kharlyngdoh J, Modig C, Olsson PE. 2014 1,2-Dibromo-4-(1,2 dibromoethyl) cyclohexane (TBECH)-mediated steroid hormone receptor activation and gene regulation in chicken LMH cells. Environ Toxicol Chem. 33(4):891-899
Pradhan A, Kharlyngdoh JB, Asnake S, Olsson PE. 2013. The brominated flame retardant TBECH activates the zebrafish (Danio rerio) androgen receptor, alters gene transcription and causes developmental disturbances. Aquat Toxicol. 142-143C:63-72.
Khalaf, H., Larsson, A., Berg, H., McCrindle, R, Arsenault, G., Olsson, P.-E. (2009) Diastereomers of the brominated flame retardant 1,2-dibromo-4-(1,2-dibromoethyl)cyclohexane induce androgen receptor activation in HepG2 and the prostate cancer cell line LNCaP. Environ. Health Perspect. 28, 1063-1071.
Olsson, P.-E., Larsson, A., Karlsson, J., Modig, C. and Mayer, I. (2008) Molecular markers of androgen disruption. In: Fish reproduction: Cytology, Biology and Ecology. Arukwe A et al. (Eds.) pp 515-534.
Hossain, M.S., Larsson, A., Scherbak, N., Olsson, P.-E., Orban, L. (2007) Zebrafish androgen receptor: Isolation, molecular and biochemical characterization. Biol. Reprod. 78, 361?369
Larsson, A., Eriksson, L.A., Andersson, P.L., Ivarsson, P. and Olsson, P.-E. (2006) Identification of the brominated flame retardant 1,2-dibromo-4-(1,2-dibromoethyl)cyclohexane as a potent androgen agonist. J. Med. Chem. 49, 7366-7372.
Olsson, P.-E., Berg, A.H., von Hofsten, J., Grahn, B., Hellqvist, A., Larsson, A., Karlsson, J., Modig, C., Borg, B. And Thomas, P. (2005) Molecular cloning and characterization of a nuclear androgen receptor activated by 11-ketotestosterone. Rep. Biol. Endocrinol. 3, 37.
Jones, I., Lindberg, C., Jakobsson, S., Hellman, U., Hellqvist, A., Borg, B. and Olsson, P.-E. (2001) Molecular cloning and characterization of spiggin: an androgen regulated extraorganismal adhesive with structural similarities to von Willebrand Factor -related proteins. J. Biol. Chem. 276: 17857-17863.
Researchers
- Solomon Asnake
- Per-Erik Olsson